Quiz Summary
0 of 67 Questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
0 of 67 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
Categories
- Not categorized 0%
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
- 22
- 23
- 24
- 25
- 26
- 27
- 28
- 29
- 30
- 31
- 32
- 33
- 34
- 35
- 36
- 37
- 38
- 39
- 40
- 41
- 42
- 43
- 44
- 45
- 46
- 47
- 48
- 49
- 50
- 51
- 52
- 53
- 54
- 55
- 56
- 57
- 58
- 59
- 60
- 61
- 62
- 63
- 64
- 65
- 66
- 67
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 67
1. Question
Which of the following methods is used in separating a mixture of palm wine and water?
CorrectIncorrect -
Question 2 of 67
2. Question
A sample of orange juice is suspected to have been contaminated with a yellow dye. Which of the following methods can be used to detect the dye?
CorrectIncorrect -
Question 3 of 67
3. Question
A salt which loses mass when exposed to air is
CorrectIncorrect -
Question 4 of 67
4. Question
A particle that contains 17 protons, 18 neutrons and 18 electrons is
CorrectIncorrect -
Question 5 of 67
5. Question
When pollen grains are suspended in water and viewed under a microscope, they appear to be in a state of constant but erratic motion. This is due to
CorrectIncorrect -
Question 6 of 67
6. Question
The atomic mass of an atom is mostly due to the mass of
CorrectIncorrect -
Question 7 of 67
7. Question
The atomic mass number of an element is 10, then the outermost shell will have ______ electrons
CorrectIncorrect -
Question 8 of 67
8. Question
Water has a rather high boiling point despite its low molecular mass because the presence of bonding.
CorrectIncorrect -
Question 9 of 67
9. Question
The type of bond between two atoms of an element with atomic number 7 each is
CorrectIncorrect -
Question 10 of 67
10. Question
The chemically unreactive group of elements is called the
CorrectIncorrect -
Question 11 of 67
11. Question
Which of the following indicates the correct increasing order of oxidizing power of the halogens
Br – I – Cl – F
Cl – F – I – Br
F – Cl – Br – I
I – Br – Cl – F
I – Cl – Br – F
CorrectIncorrect -
Question 12 of 67
12. Question
Elements in the same group in the periodic table have the same
CorrectIncorrect -
Question 13 of 67
13. Question
The following are periodic properties EXCEPT
CorrectIncorrect -
Question 14 of 67
14. Question
If the rate of diffusion of oxygen gas is I, what will be the rate of diffusion of methane whose relative molar mass is 16?
CorrectIncorrect -
Question 15 of 67
15. Question
According to the kinetic molecular theory, molecules are in random motion. This motion is greatest when a substance is changing from
CorrectIncorrect -
Question 16 of 67
16. Question
The complex salt formed when aluminum dissolves in sodium hydroxide solution is
CorrectIncorrect -
Question 17 of 67
17. Question
X acid is colorless when pure but often yellowish due to dissolved nitrogen(iv)oxide by slight decomposition of the acid. X is
CorrectIncorrect -
Question 18 of 67
18. Question
A salt derived from a strong base and a weak acid will undergo hydrolysis and give a solution which will be
CorrectIncorrect -
Question 19 of 67
19. Question
_________ of an acid is the number of replaceable Hydrogen ions in one molecule of the acid
CorrectIncorrect -
Question 20 of 67
20. Question
Which of the following salts is most conveniently prepared by neutralization method?
CorrectIncorrect -
Question 21 of 67
21. Question
Na2CO(aq) + 2HCl(aq) + 2NACl(aq) + H2O + CO2 (g) Using the equation above, what volume of carbon (IV) oxide in dm3 measured at s.t.p is liberated when 36g of sodium trioxocarbonate (IV) is dissolved in hydrochloric acid (1 mole of gas occupies 22.4dm3 at stp) [Na =23, C=12, O=16]
CorrectIncorrect -
Question 22 of 67
22. Question
If 14.8g of a salt dissolved in 25cm3 of distilled water with a concentration of 0.80 moldm-3. The molar mass of the salt in gmol-1 is
CorrectIncorrect -
Question 23 of 67
23. Question
Which of the following is a frozen planet
CorrectIncorrect -
Question 24 of 67
24. Question
The mass of carbon in grams needed to reduce 15.9of copper (II) oxide is [C=12 Cu=63.5 O=16]
CorrectIncorrect -
Question 25 of 67
25. Question
In the reaction
2Pb(NO3)2(s) 2 PbO (s) + 4NO2 (g)
CorrectIncorrect -
Question 26 of 67
26. Question
Which of the following is true of an endothermic reaction?
CorrectIncorrect -
Question 27 of 67
27. Question
Which of the following statements is correct? Rate of chemical reaction can altered by the
CorrectIncorrect -
Question 28 of 67
28. Question
Which of the following compounds will form a solution when exposed to air?
CorrectIncorrect -
Question 29 of 67
29. Question
In the electrolysis of brine, the anode is
CorrectIncorrect -
Question 30 of 67
30. Question
Which of the following reactions takes place in the cathode compartment during the electrolysis of copper (II) chloride solution?
CorrectIncorrect -
Question 31 of 67
31. Question
When the concentration of an electrolyte decreases, the conductivity
CorrectIncorrect -
Question 32 of 67
32. Question
N2O4(g) 2NO2(g). In the endothermic reaction above, more product will be formed a/an
CorrectIncorrect -
Question 33 of 67
33. Question
Calculate s in JK-1 mol -1 for the conversion of one mole of liquid water to vapour at 100C given that the heat of vaporization of water = 2260.87Jg-1
CorrectIncorrect -
Question 34 of 67
34. Question
A measure of the degree of disorderliness in a chemical system is known as the
CorrectIncorrect -
Question 35 of 67
35. Question
2KBr + Cl2 2KCl + Br2 In the reaction above, the role of chlorine is that of a/an
CorrectIncorrect -
Question 36 of 67
36. Question
Alkanol + Alkanoic acid Ester + Water . The reverse reaction of the equation is known as
CorrectIncorrect -
Question 37 of 67
37. Question
What is the product of the reaction between ethanol and excess acidized KMnO4 solution?
CorrectIncorrect -
Question 38 of 67
38. Question
Which of the following compounds is a member5 of the series with the general molecular formula CnH2n-2 ?
CorrectIncorrect -
Question 39 of 67
39. Question
Which of theses compounds will react with NaOH to form a salt?
CorrectIncorrect -
Question 40 of 67
40. Question
Alkanoates are produced from alkanols by
CorrectIncorrect -
Question 41 of 67
41. Question

The compound with the structure above is an
CorrectIncorrect -
Question 42 of 67
42. Question
What is the IUPAC name of the compound with the following structure?
CorrectIncorrect -
Question 43 of 67
43. Question
Which of the following is an allotropic form of oxygen?
CorrectIncorrect -
Question 44 of 67
44. Question
When steam is passed over white hot coke, the products are:
CorrectIncorrect -
Question 45 of 67
45. Question
Which of the following is an allotropic form of oxygen?
CorrectIncorrect -
Question 46 of 67
46. Question
Which of the following equations represents the reaction of Chlorine with hot concentrated sodium hydroxide solution?
CorrectIncorrect -
Question 47 of 67
47. Question
In the laboratory preparation of Chlorine, the drying agent used is
CorrectIncorrect -
Question 48 of 67
48. Question
Halogens are strong
CorrectIncorrect -
Question 49 of 67
49. Question
Chlorine is NOT used in
CorrectIncorrect -
Question 50 of 67
50. Question
Which of the following gases is colourless, odourless and soluble in KOH solution?
CorrectIncorrect -
Question 51 of 67
51. Question
A mixture of two immiscible liquids can be separated by
CorrectIncorrect -
Question 52 of 67
52. Question
Which of these is NOT a property of metals?
CorrectIncorrect -
Question 53 of 67
53. Question
The best way to distinguish between Na2CO3 and NaHCO3 is by
CorrectIncorrect -
Question 54 of 67
54. Question
The green colour solution of an Iron (II) salt changes to a brown colour solution of Iron (III) salt by a process known as
CorrectIncorrect -
Question 55 of 67
55. Question
Which of the following is a frozen planet
CorrectIncorrect -
Question 56 of 67
56. Question
A plastic which CANNOT be softened by heat is described as
CorrectIncorrect -
Question 57 of 67
57. Question
What type of bond exists in sodium chloride?
CorrectIncorrect -
Question 58 of 67
58. Question
A plastic which CANNOT be softened by heat is described as
CorrectIncorrect -
Question 59 of 67
59. Question
Which of the following statements is correct? Rate of chemical reaction can altered by the
CorrectIncorrect -
Question 60 of 67
60. Question
The following are divisions of chemical industries EXCEPT
CorrectIncorrect -
Question 61 of 67
61. Question
Which of the following equations represents a Beta decay?
CorrectIncorrect -
Question 62 of 67
62. Question
State TWO differences between chemical properties of metals and non-metals
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted. -
-
Question 63 of 67
63. Question
(a).i. State TWO differences between chemical properties of metals and non-metals
- List TWO methods of extracting metals from their ores
(b) i. State Avogadro’s law
i. Which state of matter contains that are
i. Readily compressed
ii. Held firmly together by some forces of cohesion?
iii. Involved in rapid random motion?
(c) i. Which of the elements represented as A to E in the table above is
i. a transition metal?
ii. an alkaline earth metal?
III. least reactive?
iv. the most electronegative
ii. Name the type of bond that exists in a compound formed when element D reacts with oxygen
(d) Write equations for the reactions at each electrode when a dilute solution of sodium chloride is electrolysed using carbon electrodes
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint

(ii) Two methods of extracting metals from their ores.
– Electrolytic reduction
– Chemical and thermal reduction
b(i) Avogadro’s law states that equal volumes of all gases
at the same temperature and pressure contain the
same number of molecules
(ii) (I) Gas
(II) Solid
(III) Gas
c(i) (I) E
(II) D
(III) A
(IV) B
(ii) Electrovalent (ionic) bond


-
Question 64 of 67
64. Question
a(i) Methane reacts with chlorine under a certain condition to produce tetrachloromethane.
i. State the condition for the reaction
ii. Name the reaction
iii. Mention TWO uses of methane
iv. Name ONE major natural source of ethanoic acid
bi. Mention ONE similarity between the reaction of ethanol with sodium and that of sodium with water
ii. Write the two structures of TWO isomers of C3H8O
iii. Differentiate between a fine chemical and a heavy chemical and cite ONE example of each.
c. Explain the following observations:
i. Crystals of washing soda become powdery on exposure to air for a long time
ii. Give the IUPAC name of the following salts: CoCl2 and Mg(NO3)2
iii. State ONE physical method and ONE chemical method in each case by which the following can be removed from water:
(a). Permanent hardness
(b). A suspension of CaCO3
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
a(i)(I) Condition for the reaction – presence of ultra-
violet light (sunlight)
(II) Chlorination reaction
(III) Two uses of methane
– Methane is used as fuel
– It is used for the production of synthesis gas
– It is used for the production of valuable
substance such as carbon black, trichloromethane
and tetrachloromethane
(Any Two)
(IV) One major natural source of ethanoic acid is vineger
b(i) One similarity between the reaction of ethanol with sodium and that of sodium with water is that hydrogen gas is liberated in both reactions.
(ii) Structures of two isomers of C3H8O

(III) Fine chemicals are chemicals produced in small
quantities for specific purposes and to a very high
degree of purity while heavy chemicals are chemicals used extensively and are produced in very large quantities.
One example of fine chemicals – drugs, dyes laboratory reagents, perfumes e.tc. (Any one)
One example of heavy chemicals – H2SO4, NaOH, CaCO3, bleaching powder, metals (Iron, Copper e.t.c)
etc. (Any One )
C(i) This is because the crystals of washing soda on exposure to air for a long time last its water of crystallization to become an anhydrous salt (powdered form). The Phenomenon is called efflorescence.
(ii) ZnO – Amphoteric oxide
CO – Neutral oxide
NO – Neutral oxide
SO2 – Acidic oxide
d(i) CoCl2 – Cobalt (II) Chloride
Mg(NO3)2 – Magnesium trioxonitrate (V)
(ii) Chlorofluorocarbons
(III) a permanent hardness
– Physical method of removal
– Permutit method i.e ion – exchange process.
– Distillation process.
(Any One)
Chemical Method – Addition of washing soda.
Addition of caustic soda.
(Any One)
- b) A suspension of CaCO3
Physical method of removal – Filtration
Chemical Method of removal – Addition of acid e.g Hydrochloric acid
-
-
Question 65 of 67
65. Question
(a) i. Draw a labeled diagram for the laboratory preparation of a dry sample of chlorine gas.
ii. Give TWO differences between conductors and electrolytes(b) Write equations to represent the reaction of chlorine gas with:
i. iron(ii) chloride solution
ii. hot concentrated sodium hydroxide solution
iii. potassium iodide solution(c) state what is observed on:
I. bubbling hydrogen chloride gas into an aqueous solution of lead trioxonitrate (v)
II. heating the mixture from c(i) above to boiling and then allow it cool.
(d) Define oxidation in terms of electron transfer.
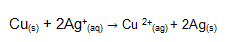
Identify the specie that is reduced in the equation above.
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
a(i) Laboratory preparation of a dry sample chlorine gas

(ii) Two differences between conductor and electrolytes

b. Reaction of chlorine gas with;
(i) Iron (II) chloride solution

c(i) A white precipitate of lead (II) chloride is formed.
(ii) On heating to boiling, the precipitate from C(I)dissolves, and when the mixture is allowed to cool, precipitate re-appears.
(II) Two laboratory reagents needed to oxidize ethanol to ethanoic acid
– Acidified potassium tetraoxomanganate (VII)
– Acidified potassium heptaoxodichromate (VI)
C(i) Esterification
(ii) Hydrolysis
(iii) Hydrogenation
(v) Saponification
d(i) Managanese (VI) oxide
(ii) Alkaline pyrogallol
-
-
Question 66 of 67
66. Question
(a) Define the term-hydrocarbons
i. A hydrocarbon consist of 92.3% carbon. If its vapour density is 39, determine its molecular formula. (H=1, C=12)
(b) Outline a suitable procedure for obtaining ethanol from cassava tubers
i. List TWO laboratory reagents needed to oxidize ethanol to ethanoic acid.
c. What name is given to each of the following processes:
i. conversion of alkanols to alkanoates
ii. breaking down of proteins to amino acids
iii. conversion of oils to fats
iv. alkaline hydrolysis of fats and oils
(c) Potassium trioxochlorate (v) undergoes thermal decomposition according to the following equation

d. What substance could be used in the laboratory to:
a. Increase the rate of the reaction
b. Absorb the oxygen produced
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
a(i) Hydrocarbons are organic compounds consisting of carbon and hydrogen only

b(1) Laboratory procedure for obtaining ethanol from cassava tubbers
– The cassava is crushed and pressure – cooked to extract starch granules (i.e mash)
– Treat the mash with malt at 50oC. the diastase in the malt converts the starch to maltose
– Add yeast to the maltose solution and leave to ferment for 1- 3 days
– There are two enzymes present in the yeast maltase and zymase
– The maltase in the yeast converts the maltose to glucose. The zymase converts the glucose to ethanol and carbon (IV) oxide
– Distil to obtain ethanol.
(II)Two laboratory reagents needed to oxidize ethanol to ethanoic acid
– Acidified potassium tetraoxomanganate (VII)
– Acidified potassium heptaoxodichromate (VI)C(i)Esterification
(ii)Hydrolysis
(iii)Hydrogenation
(v)Saponificationd (i)Managanese (VI) oxide
(ii)Alkaline pyrogallol -
-
Question 67 of 67
67. Question
(a) i. Define heat of combustion
ii. Name the container used for determining the heat of a reaction(b) i. The heat of combustion of carbon in excess air is 3935 kJ
ii. Sketch an energy profile diagram for the reaction
iii. Explain why the value of the heat of neutralization of strong acids by strong bases is constant(c) Give the reasons for the following:
i. rusting of iron is regarded as slow combustion
ii. an aqueous solution of silver trioxonitrate (v) gives a white precipitate with KCl but not with KClO3
iii. iron is better protected from corrosion by plating it with zinc than with tin
(d) When ethane 1,2 dioc acid is heated with conc. H2SO4 acid, a reaction represented by the following equation occurs
i. Name the type of process involved in the reaction
ii. What is the basicity of ethane-1,2-dioic acid?
iii. List THREE differences in the chemical properties of the two oxides produced during the reaction.
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
a (i) Heat of combustion is the heat evolved when one mole of a substance is completely burnt in oxygen
(ii) The container used for determining the heat of a reaction is calorimeterb(i)

(ii) C(s) + O2(g) CO2(g) H= – 3935kJ
I mole of C = 12g
From the equation,
12g of C evolved – 3935KJ
60g of C will evolve 60 x (-3935)
12
= – 19675KJ
(iii) Neutralization reaction can be represented as

Strong acids and strong bases ionize completely in water to produce H3O+ and OH- respectively. in all cases one mole of H3O+ of a strong acid completely neutralises one mole of OH- of a strong base. Thus the enthalpy of neutralization is constant.
c(i)Both rusting and combustion processes involve reaction with oxygen to produce oxide. both reactions are exothermic although rusting takes a longer period of time than combustion.
(ii)The white precipitate formed is silver chloride. In KCl, therefore free chloride ions in solution to form the precipitate, but in KCLO3, the chlorine
atom is present in the solution as a complex ion (radical). Thus it is not available to react with the silver ion to form the precipitate.(iii)Zinc is more reactive than iron which is more reactive than tin. thus, when a tin-plated iron is exposed, the iron goes into solution and rust
but in case of galvanized iron, it is the zinc that dissolves thereby protecting the iron better.d(i) The type of process involved in the reaction is dehydration process.
(ii) The basicity of ethane – 1, 2- dioic acid is 2
(iii) Three differences in the chemical properties of carbon (II) oxide and carbon (IV) oxide.

-


