Quiz Summary
0 of 3 Questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
0 of 3 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
Categories
- Not categorized 0%
- 1
- 2
- 3
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 3
1. Question
1. The following materials were provided for a titration experiment: Solution A which contains 10.9gdm-3 of dibasic acid H2Y, solution B containing 1.0 moldm-3 of sodium trioxocarbonate (IV), distilled water, 250cm3 pippette, and a bottle each of methyl orange and phenolphthalein indicators.
a. What would be the colour of
i. Methyl orange in solution A and B
ii. Phenolphthalein in solution A and B
iii. Which of the two indicators will be more suitable for the titration? Give reasonsb. List THREE measures to be taken during titration experiments to ensure accurate results.
c. 22.5cm3 of Solution A neutralized 25.0cm3 of 0.10moldm-3 solution of sodium trioxocarbonate (IV). Calculate the
i. Concentration of solution A in moldm-3
ii. Molar mass of the acid H2YThe equation for the reaction is Na 2 CO3
d. Name ONE reagent that is kept inside amber coloured bottle and state a reason for doing so.-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
-
-
Question 2 of 3
2. Question
The following heat tests were carried out on given samples of salts. Give the possible observations and inferences. The observation given must include the confirmatory given must include the confirmatory test

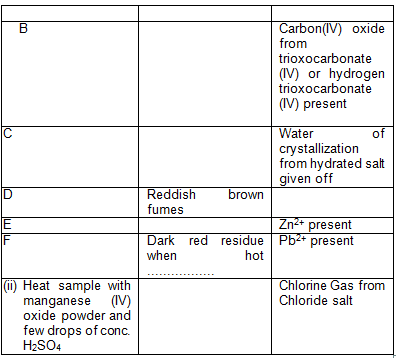
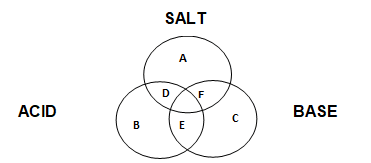
iii. Place the following substances into their correct position using the alphabets in the Venn diagram above.
i. Zn (OH) Cl
ii. CH3COOH
iii. AlCl3
iv. NaHSO4
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted. -
-
Question 3 of 3
3. Question
CorrectIncorrect


