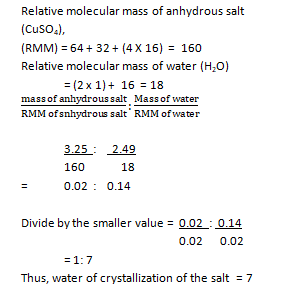chemistry neco 2013
Quiz Summary
0 of 66 Questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
0 of 66 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
Categories
- Not categorized 0%
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
- 22
- 23
- 24
- 25
- 26
- 27
- 28
- 29
- 30
- 31
- 32
- 33
- 34
- 35
- 36
- 37
- 38
- 39
- 40
- 41
- 42
- 43
- 44
- 45
- 46
- 47
- 48
- 49
- 50
- 51
- 52
- 53
- 54
- 55
- 56
- 57
- 58
- 59
- 60
- 61
- 62
- 63
- 64
- 65
- 66
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 66
1. Question
The following are properties of electrovalent compounds EXECPT that they
CorrectIncorrect -
Question 2 of 66
2. Question
The components of universal indicator solution can be separated by
CorrectIncorrect -
Question 3 of 66
3. Question
A mixture of two immiscible liquids can be separated by
CorrectIncorrect -
Question 4 of 66
4. Question
Which of the following is not an evidence for the particulate nature of matter
CorrectIncorrect -
Question 5 of 66
5. Question
How many atoms are contained in 2g of hydrogen gas?
(H=1, NA =6.02×1023 )
CorrectIncorrect -
Question 6 of 66
6. Question
Which orbital has a spherical sondhape ?
CorrectIncorrect -
Question 7 of 66
7. Question
The bonds present in ammonium ion is/are
CorrectIncorrect -
Question 8 of 66
8. Question
How many electrons are in the outermost shell of a chlorine atom?
CorrectIncorrect -
Question 9 of 66
9. Question
In the periodic table, alkaline earth metals are found in Group
CorrectIncorrect -
Question 10 of 66
10. Question

The orbital above shows the electronic configuration of a/an
CorrectIncorrect -
Question 11 of 66
11. Question
which of the following properties decreases across the period?
CorrectIncorrect -
Question 12 of 66
12. Question
The percentage of oxygen in calciumtrioxocarbonate(iv) (CaCO3) is (Ca =40, C =12, O =16)
CorrectIncorrect -
Question 13 of 66
13. Question
From xNH3(g)+ yO2(g) →zNO(g)+3H2(g) the value of x,y and z respectively are
CorrectIncorrect -
Question 14 of 66
14. Question
Which of the following is NOT a property of an acid?
CorrectIncorrect -
Question 15 of 66
15. Question
A soluble base is called
CorrectIncorrect -
Question 16 of 66
16. Question
A salt without its water of crystallization is said to be
CorrectIncorrect -
Question 17 of 66
17. Question
Which of the following pH values indicates a basic Solution
CorrectIncorrect -
Question 18 of 66
18. Question
If 25.00cm3 of 0.1moldm-3 sodium hydroxide neutralizes 21.00 cm3of hydrochloric acid, the concentration of the acid in moldm-3 is
CorrectIncorrect -
Question 19 of 66
19. Question
Calculate the volume of gas in dm3 used up when 0.5 of it is involved in a reaction. (1 mole of a gas occupies 22.4 dm3 at STP)
CorrectIncorrect -
Question 20 of 66
20. Question
Calculate the mass in gram of anhydrous sodiumtrioxocarbonate (IV) in 500cm3 of 0.1 moldm-3 of the solution (Na=23, C=12, O=16)
CorrectIncorrect -
Question 21 of 66
21. Question
Calculate the volume of gas in dm3 used up when 0.5 of it is involved in a reaction. (1 mole of a gas occupies 22.4 dm3 at STP)
CorrectIncorrect -
Question 22 of 66
22. Question
When aqueous ammonium is added to one of the following solution, a white precipitate which dissolved in excess ammonia is formed. The solution is
CorrectIncorrect -
Question 23 of 66
23. Question
Which of the following compounds will NOT when strongly heated?
CorrectIncorrect -
Question 24 of 66
24. Question
Which of the following is a condition for a spontaneous reaction?
CorrectIncorrect -
Question 25 of 66
25. Question

reaction above is
CorrectIncorrect -
Question 26 of 66
26. Question
How many faradays will be required to literate 1mole of aluminum ion from the solution during electrolysis?
CorrectIncorrect -
Question 27 of 66
27. Question
The following occur during electrolysis, EXCEPT
CorrectIncorrect -
Question 28 of 66
28. Question
The arrangement of two different metals in aqueous solution of their salts to produce an electric current is
CorrectIncorrect -
Question 29 of 66
29. Question
Calculate the rate of reaction in gmin -1 when 0.5gOf calcium trioxocarbonate(IV) was added to excess dilute hydrochloric acid for five minutes and carbon(IV) oxide was evolved
CorrectIncorrect -
Question 30 of 66
30. Question
In the equation
 ,S represent theCorrectIncorrect
,S represent theCorrectIncorrect -
Question 31 of 66
31. Question
Which of the following does NOT affect the rate of a chemical reaction?
CorrectIncorrect -
Question 32 of 66
32. Question
Equilibrium is said to be attained in a reversible reaction when
CorrectIncorrect -
Question 33 of 66
33. Question
An example of a disaccharide is
CorrectIncorrect -
Question 34 of 66
34. Question
Ethene and ethyne can be distinguished from each other by bubbling each into
CorrectIncorrect -
Question 35 of 66
35. Question
The name of the compound is
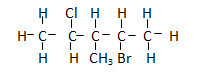 CorrectIncorrect
CorrectIncorrect -
Question 36 of 66
36. Question
What is the molecular formula of a compound whose empirical formula is CH2 O and molar mass is 180? (H=1, C=12, 0=16)
CorrectIncorrect -
Question 37 of 66
37. Question
Which of the following involves decomposition of sugar by action enzymes?
CorrectIncorrect -
Question 38 of 66
38. Question
Which of the following carbohydrate does NOT usually occur in the crystalline form?
CorrectIncorrect -
Question 39 of 66
39. Question
An alkene can be converted to an alkane by
CorrectIncorrect -
Question 40 of 66
40. Question
The alkanol with the formula (CH3)3 COH is said to be
CorrectIncorrect -
Question 41 of 66
41. Question
Which of the following compounds is a member5 of the series with the general molecular formula CnH2n-2 ?
CorrectIncorrect -
Question 42 of 66
42. Question
Which of theses compounds will react with NaOH to form a salt?
CorrectIncorrect -
Question 43 of 66
43. Question
Chlorine is used in water treatment as a/an
CorrectIncorrect -
Question 44 of 66
44. Question
Which of the following HCO3– or SO42 – cause hardness of water?
CorrectIncorrect -
Question 45 of 66
45. Question
Hydrogen is used for the following EXCEPT
CorrectIncorrect -
Question 46 of 66
46. Question
Producer gas is a mixture for the
CorrectIncorrect -
Question 47 of 66
47. Question
which of the following is not air pollutant?
CorrectIncorrect -
Question 48 of 66
48. Question
The following can be obtained directly from the destructive distillation of coal, EXCEPT
CorrectIncorrect -
Question 49 of 66
49. Question
A method that cannot be used to prevent rusting is
CorrectIncorrect -
Question 50 of 66
50. Question
Bronze is an alloy of copper and
CorrectIncorrect -
Question 51 of 66
51. Question
The following are properties of transition melting EXCEPT
CorrectIncorrect -
Question 52 of 66
52. Question
Clay is the major raw material for production of
CorrectIncorrect -
Question 53 of 66
53. Question
The major raw material in a plastic industry is
CorrectIncorrect -
Question 54 of 66
54. Question
Which of the following is NOT a detector of radio active emission?
CorrectIncorrect -
Question 55 of 66
55. Question
Changes in the physical states of substance M is illustrated below:

The phase order w, v and u represent
CorrectIncorrect -
Question 56 of 66
56. Question

The equation above illustrates
CorrectIncorrect -
Question 57 of 66
57. Question
The planet that is NOT among the solar system is
CorrectIncorrect -
Question 58 of 66
58. Question
The ideal gas equation can be represented by
CorrectIncorrect -
Question 59 of 66
59. Question
The pressure exerted by a gas is as a result of the
CorrectIncorrect -
Question 60 of 66
60. Question
The theory that electrons in an atom revolve round the nucleus was postulated by
CorrectIncorrect -
Question 61 of 66
61. Question
Define oxidation?
Clssify the following substance as oxidizing or reducing agent.
– Trioxonitrate (V) acid
– Tetraoxosulphate (VI) acid
– Chlorine
Carbon (II) oxide
(b) i. Define allotropy
- name TWO crystalline forms of sulphur
iii. Give TWO uses of sulphur
(c) i. Give the percentage composition of the following constituents of air.
– Oxygen
– Nitrogen
– Carbon (IV) oxide
– Rare gases
ii. Give the product formed by chlorine water exposed to sunlight
iii. Write a balance chemical equation for the reaction between chlorine and water
iv. State TWO uses of chlorine
v. Give FOUR products of fractional distillation of crude oil.
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
1a(i) Oxidation is a process involving loss of
electrons
(ii) Trioxonitrate (V) acid – Oxidizing agent
Tetraoxosulphate (VI) acid – Oxidizing agent
Chlorine – Oxidizing agent
Carbon (II) oxide – Reducing agent
b(i) Allotropy – is the existence of two or more
different forms of an element in the same
physical state.
(ii) Two crystalline forms of sulphur are rhombic
sulphur and monoclinic sulphur.
(iii) Two uses of sulphur are
– Sulphur is used in the vulcanisation of rubber
– It is used in the manufacture of the bleaching
agent used in pulp and paper industry
– It is used for the production of carbon disulphide
skin ointments and dyes
– It is used to produce sulphur (IV) oxide for
manufacturing tetraoxosulphate (Vi) acid.
(Any Two)
C(i) Oxygen – 21%
Nitrogen – 78%
Carbon (IV) oxide – 0.03%
Rare gases – 1%
(ii) The products formed by chlorine water when
exposed to sunlight are hydrochloric acid and
oxygen.
(iii) Cl2(g) + H2O(l) HCl(aq) + HOCl(aq)
- iv) Two uses of chlorine
– Chlorine is used in the sterilization of water for
domestic and industrial uses.
– It is used in the treatment of sewage
– It is used as a bleaching agent for cotton, linen
and wood – pulp
– It is used in the manufacture of important organic solvents such as trichloromethane, tetrachloromethane e.t.c
(v) Four products of fractional distillation of crude oil:-
– Petroleum gas e.g methane, ethane, propane e.tc
– Petrol (or gasoline)
– Kerosene
– Gas oil and diesel oil
– Lubricating oils
-
Question 62 of 66
62. Question
(a) i. Define the term hard water.
- Mention the types of hard water
iii. Name ONE substance each that causes the hardness of water in 2a(ii).
- Give ONE method each for softening each type of the hardness in 2a(ii)
(b) i. Name the THREE radioactive emissions.
- State ONE difference between nuclear fusion
(c) i. Write the structural formula of:
– 2,3,4 – trimethylpentane
– 2 – bromo-3-methylpentan-1-ol
Trans 2, 3-dichlorobut-2-ene
ii. Write a chemical equation that illustrates the oxidation of:
(i) A secondary
(ii) A primary alkanol
State One physical property of:
(i) Diamond
(ii) Graphite
d. i. Define an oxidizing agent?
ii. Give ONE example of an oxidizing agent
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
a)(i) Hard water is defined as the water which
does not form lather readily with soap
(ii) Types of hard water -Temporarily hard water
– Permanently hard water
(iii) Temporary hardness is caused by the
presence of dissolved calcium hydrogentrioxocarbonate (iv) [Ca(HCO3)2]
Permanent hardness is caused by the
presence of calcuim tetraoxosulphate (VI), magnesium tetraoxosulphate (VI), Calcium
Chloride or magnesium chloride.
(IV) One method of softening temporary
hardness :
– It can be removed by boiling
– It can be removed by adding calculated amount of
slaked lime [Ca(OH)2)
(Any One)
One method of softening permanent hardness
– It can be removed by the addition of washing soda
– It can be removed by the addition of caustic soda
– It can be removed by the addition of permutit (or zeolite)
– It can be removed by distillation
(Any One)
b(i) The three radioactive emissions are
– Alpha emission, Beta emission and gamma radiation
(ii) One difference between nuclear fission and nuclear fusion
– Nuclear fission involves the splitting of heavy nucleus into two large parts while nuclear fusion involves the fusing of two or more smaller nuclei to form a large nucleus.
– Nuclear fission requires a neutron to start the reaction while nuclear fusion does not require neutron to start.
– Nuclear fission produces three neutrons in each process while nuclear fusion produces one neutron.
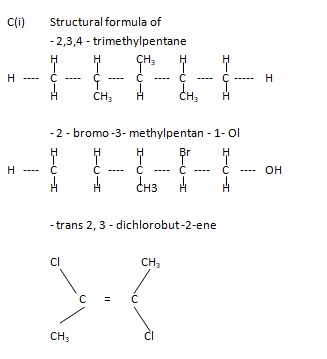
Oxygen, Manganese (IV) oxide, Hydrogen
peroxide,
acidified potassium tetraoxomanganate (VII), Chlorine etc
-
Question 63 of 66
63. Question
(a) i. State THREE factors that affect selective discharge
of ions in solution during electrolysis
- State THREE uses of electrolysis
iii. Calculate the mass of silver deposited when a current 2.6 amperes is passed through a solution of silver for 20 minutes. (Ag = 108; IF = 96500C)
(b) i. List TWO conditions required for rusting to take place.
- Mention TWO differences between rusting and burning
(c) i. Define covalent bond.
- Give THREE properties of covalent compounds
iii. Mention the type of bond that exists in compound NH4Cl
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
(i) Three factors that affect selective discharge of ions in solution during electrolysis are
– Position of ions in the electrochemical series
– The concentration of the ions in the electrolyte and
– The nature of the electrode.
(ii) Three uses of electrolysis
– it is used for purification of metals e.g Cu, Hg etc
– It is usede for electroplating of one metal by another
– It is used for the extraction of elements
– It is used for the preparation of certain important
compounds such as sodium hydroxide and sodium
trioxochlorate (V)

b(i) Two conditions required for rusting to take
place are moisture and oxygen (or air)
(ii) Two differences between rusting and burning
– Water is necessary for rusting to take place
while burning is difficult in the presence of water
– Burning is an active rapid process while rusting
is a very slow process
– The heat given out during burning is
released in large amount within a short
period of time and is easily detected while d heat given out during rusting is released in
small amount over a long period of time
and is not perceptible
C(i) Covalent bond is a bond formed by the
sharing of a pair of electrons between two
reacting atoms
(ii) Three properties of covalent compounds
– Covalent compounds consist of molecules with
definite shapes.
– They are gases or volatile lequids at room temperature.
– They have low melting and boiling points
– They readily dissolve in non-polar organic solvents such as benzene, toluene e.tc.
– They do not conduct electricity i.e they are non
electrolytes
(Any three)
(iii) The types of bond that exist in NH4Cl are ordinary covalent bond, Co-ordinate covalent (dative) bond and ionic bond.
-
Question 64 of 66
64. Question
(a) i. State TWO differences between chemical reaction and nuclear reactions.
- Explain the term esterification, using chemical equations only
iii. Define activation energy
(b) Use the information in the below to answer questions i – iv.

Which of the atom(S)
- are isotopes of the same elements?
- Contains 18 neutrons?
- Is chemically unreactive?
- Forms ionic bond with C?
- Belongs to S-block in the periodic table?
(c) i. What does each symbol represent in the expression below?

Give ONE ore of the following metals
– Aluminum
– Iron
iii. For each of the ores named in d(ii) above, list the method of extraction.
- 50cm3 of hydrogen is sparked with 20cm3 of oxygen at 100oC and 1 atmosphere. What is the total volume of the residual gases?
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
a(i) Two differences between chemical reactions and nuclear reactions

20cm3 of O2 (2 x 20)cm3 of H2O(g)
= 40cm3
volume of unused H2 = (50 – 40)cm3 = 10cm3
Total volume of the residual gases
= Volume of unused H2 + volume of steam(H2O)
formed
= 10 + 40 = 50cm3
(ii) Esterification, using chemical equations only

(iii) Activation Energy: is the minimum amount of energy required for chemical reaction to occur
b(1) A and D
(ii) C
(iii) E
(iv) B
(v) B
C(i) A represents Mass number
Z represents Atomic number
(ii) One ore of
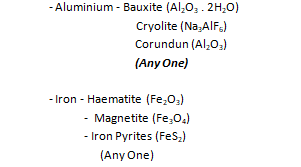
(iii) Aluminium – it is extracted by the electrolysis of the molten oxide.
Iron – it is extracted in a blast furnace by chemical and thermal reduction of its ores.

[N2][H2]3
d(i) First Law of thermodynamics states that energy may be converted from one form to another but it cannot be created or destroyed.
(ii) Valency of an element is the combining power of an
element.

-
Question 65 of 66
65. Question
(a)i. State the role each of the following substances in the treatment of water for town supply.
- – Chlorine
– Alum
– Sand-Bed
(b) i. Give three major uses of H2SO4
- Explain the following observation: A strip of blue litmus paper dropped into concentrated H2SO4 becomes charred whereas in dilute H2SO4 it turns red and is not charred
iii. Write an equation to show the reaction of conc. H2SO4 with zinc
(c) N2(g) + 3H2(g) 2NH3(g) H = – ve
Using the expression above;
- Name the type of reaction involved
- What condition will favour the forward reaction?
iii. Mention the catalyst used
- Write the expression for equilibrium constant (Kc) of the reaction.
(d) i. State the first law of thermodynamics
- Define the valency of an element
iii. Calculate the mass of trioxonitrate (V) acid present in 50cm3 of 0.5 moldm – 3 of the solution
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
a(i) Chlorine – its role is to kill the germs in the water
Alum – Its role is to cause coagulation or
flocculation of the impurties in the water.
Sand – Bed – Its role is to cause filtration to
remove particles of dirt.
b(i) Three major uses of H2SO4
– It is used in the manufacture of fertilizers
– It is used in the manufacture of pigments
for paints
– It is used for making cellulose film, fabrics
and plastics
(ii) The Litmus paper becomes charred because of the
dehydrating property of H2SO4. As a dehydrating agent, H2SO4 removes the elements of water from the paper, thereby making it to become charred.
On the other hand the blue litmus paper turns red due to the acidic property of H2SO4. As an acid, H2SO4 turns the blue litmus paper red.
(iii) An equation to show the reaction of Conc.
H2SO4 with zinc :-
Zn(s) + 2H2SO4(aq)ZnSO4(aq) + 2H2O(l) + SO2(g)
C(i) Exothermic reaction
(ii) Low temperature (450oC) and high pressure (200atm)
(iii) Finely divided iron mixed with alumina
(iv) Kc = [NH3]2
[N2][H2]3
d(i) First Law of thermodynamics states that energy may be converted from one form to another but it cannot be created or destroyed.
(ii) Valency of an element is the combining power of an
element.
-
Question 66 of 66
66. Question
(a) i. Classify the following as organic or inorganic acid: trioxonitrate (V) acid, trioxocarbonate (IV) acid ethanedioic acid amino acid
ii. Give TWO properties of acids
iii. Mention THREE types of salt and one example of each.
(b)i. Define hygroscopy
- 5.74g of hydrated copper (II) tetraoxososulphate (VI) salt were heated of a constant mass of 3.25g. Calculate the water of crystallization of the salt. [Cu = 64, S = 32, O = 16, H = 1]
iii. Define the entropy of a system
What will be the state of the reaction when of a reaction is
i. Positive?
ii. Negative?
iii. Zero?
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted.Hint
a(i) tioxonitrate (V) acid – Inorganic acid
Trioxocarbonate (IV) acid – Inorganic acid
Ethanoic acid – Organic acid
Amino acid – organicv acid
(ii) Two properties of acids
– Acids turn moist blue litmus paper red
– Acids have sour taste
– Acids react with trioxocarbonates (IV)salts to yield
carbon (iV) oxides
– Concentrated form of strong acids are corrosive
– Acids react with active metals to yield hydrogen.
(Any two)
(iii) Three types os salts and one example of each
– Normal salt – example is sodium chloride
– Acid salt, e.g sodiumhydrogentrioxocarbonate (iv)
– Basic salt e.g zinc choride hydroxide
– Double salt e.g – Ammonium iron (II)
tetraoxosulphate (vi) hexahydrate
(Any Three)
b(i) Hygroscopy is a phenomenon whereby
substances absorb mixture on exposure to
the atmosphere to become sticky if solid
and diluted if liquid
(ii) Mass of water = mass of hydrated salt – mass of
anhydrous salt
= 5.74 – 3.25
= 2.49g
Relative molecular mass of anhydrous salt
(CuSO4),