Quiz Summary
0 of 8 Questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
0 of 8 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
Categories
- Not categorized 0%
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 8
1. Question
(a) Explain the condition of chemical equilibrium using the hypothetical equilibrium equation:
aA + bB ⇋ cC dD
[3 marks]
(b) Write the equilibrium constant, Kc expressions for the following reactions
i 3C2H2 g ⇌C2H2g 1 markii 2NOCI ⇌2NO 1 markiiiN2N4 g ⇌2NO2g 1 mark
(c) A mixture of 0.003 mol of H2 and 0.002 mole of I2 were reacted to attain equilibrium in a 2- L container at 400 oC. Analysis of the equilibrium mixtures shows that the concentration of HI is 0.0022 M. Calculate Kc for the reaction at 400 oC. [4 marks]
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted. -
-
Question 2 of 8
2. Question
Crude oil is the principal source of hydrocarbons. The following are examples of such hydrocarbons.
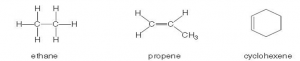
(a) Give the structural formulae of the organic products in the following reactions.
i. The reaction of ethane with bromine in the presence of U.V. light. [1 mark]
ii. The reaction of HBr with butene. [1 mark]
iii. The reaction of cyclohexene with hydrogen bromide. [1 mark]
(b) Write equations for the following reactions.
i. The complete combustion of propane. [1 mark]
ii. The action of steam on propene in the presence of a catalyst. [1 mark]
iii. The reaction of cyclohexene with hydrogen in the presence of a catalyst. [1 mark]
(c) Write a structural formula for each of the following and indicate whether it is primary, secondary or tertiary.
(i) 3 – pentanol
(ii) 2,2 – dimethyl -1- propanol
(iii) 1 – methyl cyclopentanol
(iv) 2 – methyl -2- propanol [4 marks]
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted. -
-
Question 3 of 8
3. Question
(a) Explain the following terms:
- Precision
- Accuracy. (4marks)
(b) The concentration of Nickel in Nigerian coin was determined with visible spectrophotometer, and the following results (%) were obtained: 3.65, 4.11, 3.59, 7.51, 3.95, 3.87, 4.06, 1.48, 3.60, 3.76 and 3.99. if the true concentration (%) of nickel in coin as determined by atomic absorption and inductively coupled plasma atomic emission spectrophotometer was 3.92.
Use the above data to determine:
- Absolute error
- Percentage relative error
- Average error (6 marks)
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted. -
Question 4 of 8
4. Question
(a) Name the strongest type of intermolecular force between hydrogen fluoride molecules and draw a diagram to illustrate how two molecules of HF are attracted to each other. In your diagram show all lone pairs of electrons and any partial charges. Explain the origin of these charges. Suggest why this strong intermolecular force is not present between HI molecules. [3 marks]
(b) Crystals of sodium chloride and of diamond both have giant structures. Their melting points are 1074 K and 3827 K, respectively. State the type of structure present in each case and explain why the melting point of diamond is so high. [3 marks]
(c) Write a Lewis structure and identify the octet-rule exception for (i) SClF5 (ii)BFCl2 [4 marks]
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted. -
-
Question 5 of 8
5. Question
(a) State Gay-Lussac’s law [2 marks]
(b) State Afbau’s principle and Hund’s rule of maximum multiplicity. [2 marks]
(c) what do you understand by a Bronsted acid? [1 mark]
(d) briefly explain the term “Inert Pair Effect” [2 marks].
(e) Compare and contrast the following properties of the transition elements with that of S and P- block elements. (i) Colour (ii) Atomic / ionic radius (iii) oxidation states. [3 marks]
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted. -
-
Question 6 of 8
6. Question
(a) Write an equation for the synthesis of hydrogen gas by the reaction of methane with steam. [1 mark]
(b) Compare the physical and chemical properties of the hydrides of; Na, C, N and S (3 Marks)
(c) Explain using equation why rain falling in unpolluted air is acidic with a pH of about 5.6 (2 Marks)
(d) Acid rain has a pH value less than 5.6. Explain using equation how the burning of coal can contribute to acid rain formation (2 Marks)
(e) What are the consequences of global warming on the environment? (2 Marks)
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted. -
-
Question 7 of 8
7. Question
An organic compound X contains 40% carbon, 53.3% oxygen and 6.7% hydrogen. The vapor density of X is 30. The organic compound reacted with sodium hydrogen trioxocarbonate (IV) and a colorless gas was given off which turned lime water milky. On boiling under reflux, the mixture of X and ethanol, a sweet-smelling liquid Y was formed.
(a) Determine the (i) empirical and (ii) molecular formula of X. (4 Marks)
(b) What is the name of the colourless gas given off? (1 Mark)
(c) Give the formula of the functional group present in X (1 Mark)
(d) Deduce the structure of the compound X (1 Mark)
(e) Name the reaction which took place between X and ethanol, and write an equation for the reaction between X and ethanol (3 Marks)
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted. -
-
Question 8 of 8
8. Question
(a).State three (3) major factors, which affect the discharge of ions at the electrodes during electrolysis (3 Marks)
(b) Explain why a tin-plated iron container corrodes faster than a galvanized iron container with reference to the electrochemical series (2 Marks)
(c) 528g of a trivalent metal is deposited when a current of 1.56A is passed through a solution of its salt for 1hour; calculate the relative atomic mass of the metal. Hence identify the metal (1F = 96500C) (2 Marks)
(d) State any three (3) common applications of electrolysis (3 Marks)
-
This response will be reviewed and graded after submission.
Grading can be reviewed and adjusted.Grading can be reviewed and adjusted. -









Responses