Quiz Summary
0 of 23 Questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
0 of 23 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
Categories
- Not categorized 0%
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
- 22
- 23
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 23
1. Question
Which of the following changes will favour the formation of the products of the reaction represented above, at equilibrium?
CorrectIncorrect -
Question 2 of 23
2. Question
An element \( \;\;\scriptsize {X^{226}_{88}}\) undergoes radioactive decay by emitting two alpha particles and a beta radiation. Which of the following nuclei correctly describe the product formed by the reaction?
CorrectIncorrect -
Question 3 of 23
3. Question
Which of the following Alkanols will not undergo oxidation reaction by acidified K2Cr2O7?
CorrectIncorrect -
Question 4 of 23
4. Question
The electronic configuration of the species underlined as in the molecule H2S is
CorrectIncorrect -
Question 5 of 23
5. Question
The functional group(s) of an amino acid is/are
CorrectIncorrect -
Question 6 of 23
6. Question
A student while trying to identify two gases labeled A and B, found that gas. A is acidic to litmus paper and turn acidified potassium dichromate solution green, while gas B turns red litmus paper blue and forms dense white fume with hydrogen chloride. The correct identity of A and B respectively are
CorrectIncorrect -
Question 7 of 23
7. Question
Which of the following titrations will have a solution with a pH greater than 7 at the end point
(equivalent point) of the titration?CorrectIncorrect -
Question 8 of 23
8. Question
70cm3 of hydrogen are sparked with 25cm3 of oxygen at S.T.P. The total volume of the residual gas is
CorrectIncorrect -
Question 9 of 23
9. Question
A solution X, on mixing with AgNO3 solution, gives a white precipitate soluble in NH3. A solution Y when added to X, also gives a white precipitate which is soluble on boiling. Solution Y contains
CorrectIncorrect -
Question 10 of 23
10. Question
Which of the following statement is an exception in the assumptions of the kinetic theory of gases?
CorrectIncorrect -
Question 11 of 23
11. Question
A saturated solution of AgCl was found to have a concentration of
1.30× 10-5moldm-3 The solubility product of AgCl therefore is
CorrectIncorrect -
Question 12 of 23
12. Question
0.06g of a hydrocarbon occupies 32cm3 at S.T.P. It’s formula is
CorrectIncorrect -
Question 13 of 23
13. Question
Which of the following is a redox reaction?
CorrectIncorrect -
Question 14 of 23
14. Question
Which of the following processes leads to increase in entropy?
CorrectIncorrect -
Question 15 of 23
15. Question
Which one of the following statements is true?
CorrectIncorrect -
Question 16 of 23
16. Question
A liquid begins to boil when
CorrectIncorrect -
Question 17 of 23
17. Question
An element which exists in more than one crystalline form is said to exhibit
CorrectIncorrect -
Question 18 of 23
18. Question
The electronic configuration in the ground state of the chloride ion (Cl–) is
CorrectIncorrect -
Question 19 of 23
19. Question
Which of the following is an example of chemical change?
CorrectIncorrect -
Question 20 of 23
20. Question
The oxidation number of oxygen in BaO2 is
CorrectIncorrect -
Question 21 of 23
21. Question
Which ONE of the following statements is true for the p-block elements?
CorrectIncorrect -
Question 22 of 23
22. Question
The general formula for carboxylic acids is
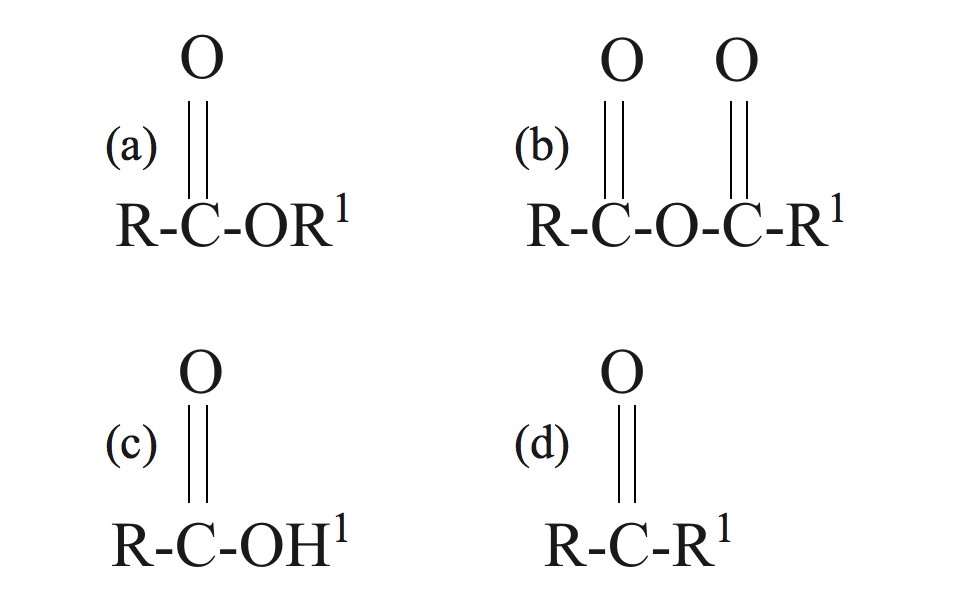 CorrectIncorrect
CorrectIncorrect -
Question 23 of 23
23. Question
The functional group represented in the compound CH3-CH2-CH2-CH2-CH=O is
CorrectIncorrect



Responses