Topic Content:
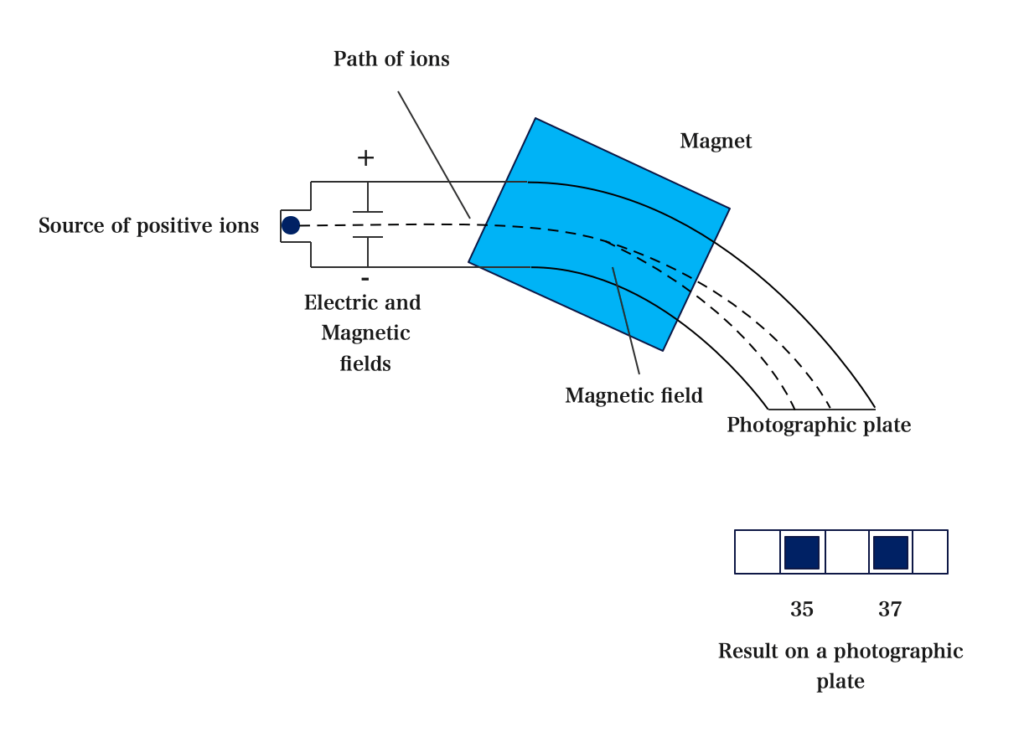
- Mass Spectrometer
Mass Spectrometer is an instrument used in measuring masses of various isotopes present in an atom.
In 1919, a chemist, Francis. W. Aston, designed an instrument called a mass Spectrometer for separating ions of different masses.
You are viewing an excerpt of this Topic. Subscribe Now to get Full Access to ALL this Subject's Topics and Quizzes for this Term!
Click on the button "Subscribe Now" below for Full Access!
Subscribe Now
Note: If you have Already Subscribed and you are seeing this message, it means you are logged out. Please Log In using the Login Button Below to Carry on Studying!



Responses