Topic Content:
- Determination of Specific Latent Heat of Fusion of Ice
Determination of Specific Latent Heat of Fusion of Ice:
The apparatus for this experiment is shown below:
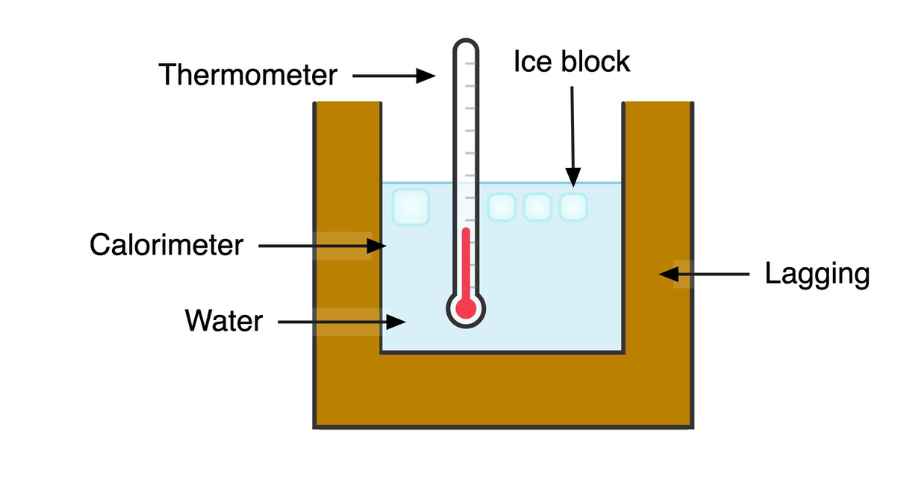
(i) A clean empty calorimeter is weighed and recorded as M1 (kg)
(ii) The calorimeter is half filled with water and weighed again as M2 (kg)
(iii) The water in the calorimeter is warmed for a few minutes to about 5ºC above room temperatureIf something is at room temperature, its temperature is neither hot nor cold. It is a range of air temperatures that most people prefer for indoor settings, usually considered to be... More. The exact temperature is recorded using a thermometer as θ1.
You are viewing an excerpt of this Topic. Subscribe Now to get Full Access to ALL this Subject's Topics and Quizzes for this Term!
Click on the button "Subscribe Now" below for Full Access!
Subscribe Now
Note: If you have Already Subscribed and you are seeing this message, it means you are logged out. Please Log In using the Login Button Below to Carry on Studying!



Responses