
Group 2 elements are called alkaline earth metals. They include magnesium, calcium, strontium, and barium.
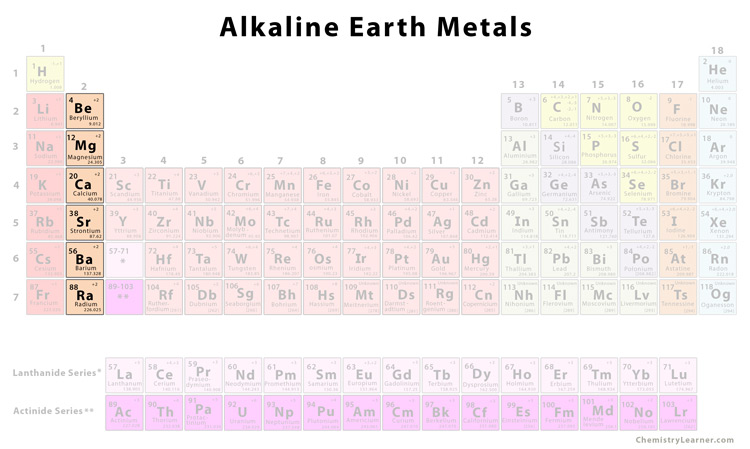
These elements have two electrons in the valence shell of their atoms and form the oxidation state of +2 in their compounds. They are good reducing agents. Alkaline earth metals have smaller atomic and ionic radii compared to the corresponding members of the alkali metals.
On moving down the group, the radii increase due to a gradual increase in the number of the shells and the screening effect. Alkaline earth metals are less reactive than alkali metals. However, reactivity increases down the group of the elements. Alkaline metals form mainly electrovalent bondsAn electrovalent or ionic bond is formed when a metal atom transfers one or more electrons to a non-metal atom. Ionic or electrovalent compounds are those compounds which are formed by... More.
1. Melting and Boiling Points: The melting and boiling point of alkaline earth metals are characteristically low and do not show regular trends down the group.
2. Ionization EnergyEnergy is the ability to do work. Energy exists in several forms such as heat, kinetic or mechanical energy, light, potential energy, and electrical energy. Units of Energy: The SI unit... More: Due to the fairly large size of the atoms, alkaline earth metals have low ionization energies which decreases down the group as atomic size increases.
3. DensityDensity is the measurement of how tightly a material is packed together i.e. how closely the particles are packed in the material. The tighter the material is packed the more its... More: Alkaline earth metals are denser and harder than the alkali metals of group-1.



Responses