When light falls on certain metal surfaces, electrons are emitted. This is known as photoelectric effect. Photoelectric emission is the giving off of electrons from a metal surface when light of appropriate frequency falls on the surface of the metal.
Findings show that the maximum kinetic energyEnergy is the ability to do work. Energy exists in several forms such as heat, kinetic or mechanical energy, light, potential energy, and electrical energy. Units of Energy: The SI unit... More of emitted electrons is directly proportional to its frequency or wavelength when maximum energy is plotted against frequency of incident light for a given metal.
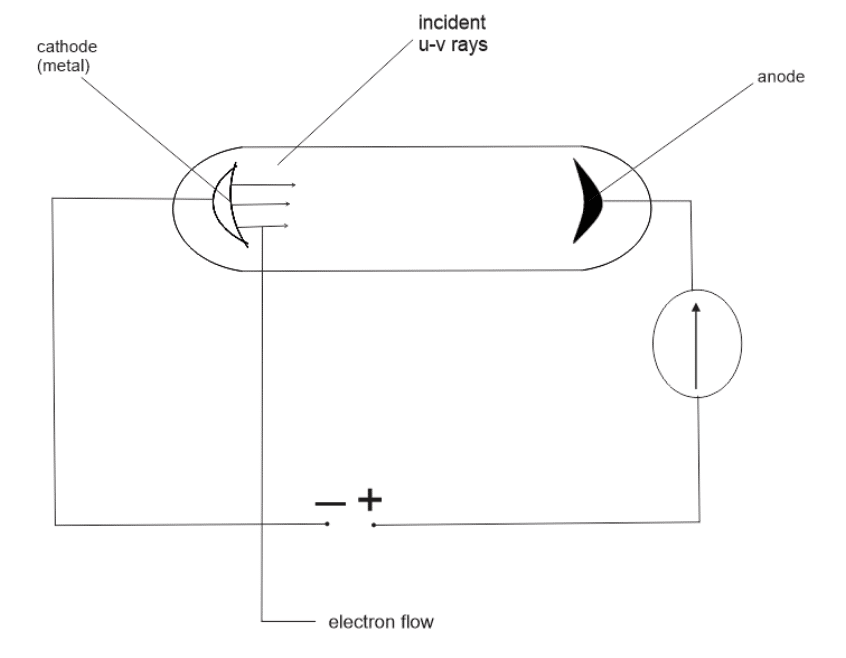
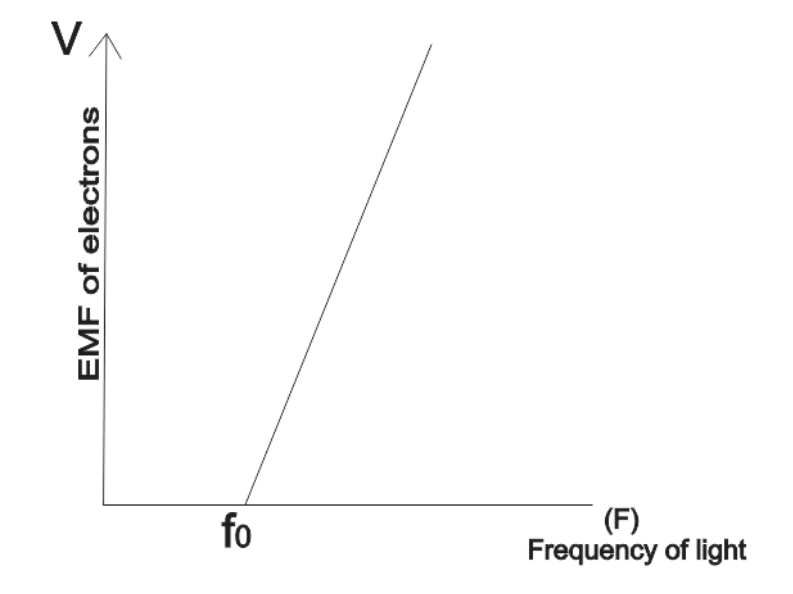
The higher the frequency, the faster electrons are removed from the metal.
Threshold frequency: This is the minimum frequency of an incident light needed to remove an electron from a metal surface.



Responses